FRONTIERS IN MEDICAL DEVICE REPROCESSING
August 2019 Innovative Health Newsletter
The PENTARAY® NAV eco high-density mapping catheter
In June, Innovative Health received clearance from FDA to reprocess and market Biosense Webster’s PENTARAY® NAV eco high-density mapping catheter. This clearance changes both the economic equation for the EP lab – and commonly held perceptions of what reprocessing can and cannot do. When Biosense Webster launched the PENTARAY as an improved alternative to the LASSO® circular mapping catheter, many labs saw their cost increase from approximately $1,250 to $1,900 per catheter – and they lost savings of $600 or more per case, because PENTARAY wasn’t available through reprocessing. With this clearance, they can now recover that loss, and savings from reprocessing diagnostic ultrasound catheters and mapping catheters can now exceed $2,000 per procedure. At the same time, Innovative Health has broken through what most have seen as a barrier to single-use device reprocessing: The ability to clean micro lumens. Innovative Health’s engineering team has worked diligently with FDA over the past several months, and cleared processes using standards more conservative than the industry has ever seen. Healthcare Purchasing News published a story about our clearance. You can also read more about it here and watch a short video here.
Heart Rhythm Society
In May 2019, Innovative Health participated in the Heart Rhythm Society conference in San Francisco. Our main message that Electrophysiology (EP) labs need to start using reprocessed sensor-enabled catheters produced a lot of good dialogue. EP labs that utilize reprocessed EP catheters, sheaths, reusable cables and specialty catheters find that they can quadruple their savings by adding diagnostic ultrasound catheters, mapping catheters and other sensor-enabled catheters. However, the largest EP device original manufacturer is doing everything it can to prevent EP labs from increasing their savings above the base level, including asserting that reprocessed devices do not have the right specifications – however they do not provide any data to back up their statements… Aaron DeTate, who manages the labs at Torrance Memorial, was with us in Innovative Health’s booth. He was able to explain how he has successfully managed to control the original device manufacturers and optimize his reprocessing savings. See brief video from the conference here.
What’s going on in Electrophysiology?
As a follow-up to the conference, Innovative Health had the opportunity to share in the press where we think the EP industry is going. “Electrophysiology, which addresses atrial fibrillation (AF) and other common heart conditions, is among the fastest growing and most expensive in the hospital’s portfolio of service lines. When hospitals struggle with
the math due to the high cost of new innovations, fewer patients will ultimately have access to the benefits of those new technologies. In EP, the clinical success of scientific advancements is directly related to the cost pressure hospitals experience. […] The future success of EP has to come with new technologies that are not just better, but also reduce procedure time, enable hospitals to cut costs and, of course, produce better patient outcomes.” See the articles in Healthcare Purchasing News, Diagnostic & Interventional Cardiology, and DOTmed Business News.
FDA Clearances and reprocessing technology
Less than a week after FDA’s clearance of PENTARAY, Innovative Health received another FDA clearance to reprocess and market the Webster Duo-Decapolar EP Catheter. This is Innovative Health’s 28th clearance.
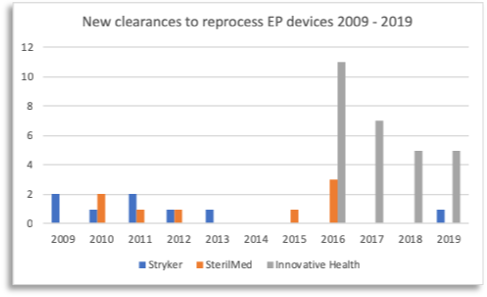
Figure 1: Comparison of reprocessors and cleared submissions
In comparison, our closest competitors SterilMed and Stryker have almost stopped getting applications cleared in the Electrophysiology space (refer to Figure 1 above). Since EP lab savings depend on new devices constantly getting cleared by the reprocessor, savings are going down. Innovative Health is now able to offer EP labs more new reprocessed devices than anyone else – bringing higher savings to EP labs. Innovative Health currently has four (4) 510(k) submissions under review with FDA. Don’t forget to ask the other reprocessors what they have in their pipeline.
Specialty Reprocessing vs. Discount Device Sales
“Many EP labs across the country rely on us to reduce procedure costs. As a specialty reprocessor, Innovative Health is able to focus on a specific clinical area, develop deep clinical and scientific competencies, and work with FDA on our submissions to achieve clearance and make higher savings available.” Innovative Health CEO Rick Ferreira said in a recent Healthcare Purchasing News article. Increasingly, getting FDA clearances that can increase EP lab savings require that reprocessors handle complex technologies and frequently develop and establish new standards and methodologies that have never been used before, in reprocessing or in the medical device industry broadly. Because of this, legacy reprocessors have a hard time accessing new technologies, and the industry is seeing the emergence of specialty reprocessors – like Innovative Health.
Hospitals increasing savings
Thanks to new clearances that allow our EP lab partners to save additional dollars in EP procedures, many of our customers have seen great results from their reprocessing programs in the first half of 2019. Innovative Health provided One East Coast hospital a $750,000 annual savings guarantee, then saw savings in excess of $1M. Other hospitals across the country that build out their reprocessing program with diagnostic ultrasound and mapping catheters are seeing device savings per procedure double. Our successful hospital partners are able to increase their savings when a) Clinicians are closely aligned with cost reduction objectives; b) EP lab management controls the influence of manufacturer representatives in the EP lab; and c) They maintain a focus on science and facts when managing their reprocessing program.
Continued threat to reprocessing savings from original device manufacturers
The biggest barrier to success in EP reprocessing remains attempts by the largest original device manufacturer to constrain hospital savings. During the past months, Biosense Webster has intensified its intimidation tactics directed at EP lab management with letters that threaten to withdraw case support during cases where reprocessed devices are used – other than those from Johnson & Johnson’s wholly owned SterilMed. The rationale for this threat is a) that Biosense Webster “doesn’t have knowledge about devices reprocessed by third parties”; b) that reprocessed devices do not have the same specifications and safety as new devices; and c) that they cannot vouch for the functionality of reprocessed devices. They are pointing to their own SterilMed division as a more appropriate reprocessing partner. While this makes sense from a business perspective, it is based on a faulty premise and designed entirely to limit reprocessing savings (SterilMed does not offer reprocessing savings of the same magnitude as Innovative Health): FDA’s clearance process ensures that reprocessed devices are substantially equivalent to new devices and pose no added patient risk. In other words, Innovative Health would be unable to obtain FDA clearance unless we could demonstrate to FDA that specifications, performance and safety are equivalent to new devices. We have seen no data to back up the claims about differences in specifications or performance. At the same time, SterilMed, Johnson & Johnson’s reprocessor, often points to the ability of FDA’s 510(k) clearance process to ensure safety and functionality of a reprocessed device is substantially equivalent to the new (predicate) device. Per the FDA, “A 510(k) is a premarket submission made to FDA to demonstrate that the device to be marketed is at least as safe and effective, that is, substantially equivalent, to a legally marketed device” (i.e. original manufacturer device). ALL reprocessors are required to demonstrate safety and efficacy in a 510(k) submission and the FDA clearance process is the same.
However, there is a different reason why Biosense Webster’s intensified focus on SterilMed is a game-changer for hospitals: Prior to the launch of PENTARAY, labs doing business with Biosense Webster had a positive impact from their SterilMed reprocessing program: SterilMed offered reprocessed LASSO catheters. With the launch of the reprocessed PENTARAY, labs working with Biosense Webster/SterilMed are effectively seeing increasing procedure costs compared with Innovative Health partners, since SterilMed does not offer reprocessed PENTARAY (or DECANAV) mapping catheters. See more about Biosense Webster’s position here and here.
